CAUTION: Investigational device. Limited by Federal (or United States) law to investigational use.

BAGUERA® C
Cervical disc prosthesis
BAGUERA® C is proven to preserve long-term mobility and function in the cervical spine. Designed in Switzerland, BAGUERA® C is intended to treat patients suffering from symptomatic cervical disc disease at one or two adjacent levels between C3 and C7.
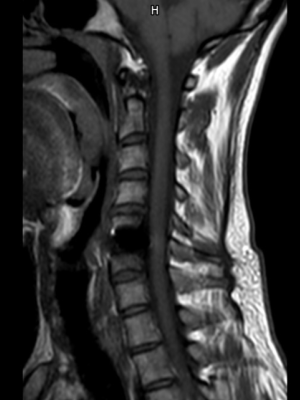
Reduced MRI Artifact
The titanium plates, coated with Diamond-Like-Carbon (DLC) reduce artifacts under MRI for a better postoperative follow-up.
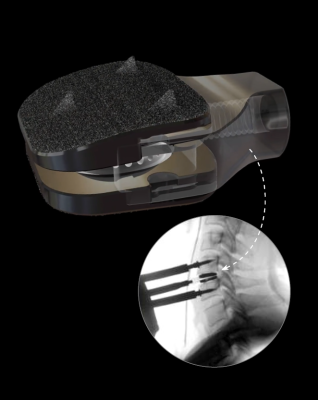
Radiolucent holder
The radiolucent holder allows for both verification of the anterior position of the device and confirmation of the fitting accuracy. Thanks to this holder, the device is delivered pre-assembled for better handling.
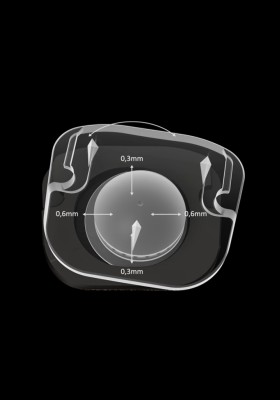
Guided mobile nucleus
The guided mobile PE nucleus is designed to prevent excessive constraints on the facet joints.
It allows 6 degrees of freedom.
Better stress distribution
Significantly lower contact pressure distribution on polyethylene central core.

About BAGUERA® C
You may also need
Our team is here to help
Contact us for more information or schedule a meeting with one of our sales representatives to explore how our products can meet your needs.